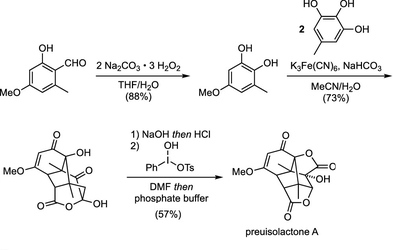
| Natural products featuring multiple stereocenters and complex structures are attractive targets for chemical synthesis because of the challenge presented in their construction. While classical total synthesis may require several steps including the addition and removal or protecting groups, biomimetic approaches offer more elegant and streamlines approaches. Novak et. al. have presented a biomimetic route towards Preuisolactone A which constructs the highly complicated molecule in 3 steps, which would normally require an estimated ~20 steps using classic methods. The key to this synthesis involves an oxidative cascade of several reactions in a single step. This synthesis serves as a probe into the biosynthesis of Preuisolactone A, suggesting that it is a polyketide formed through oxidative dimerization of a catechol with pyrogallol. |
|
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2021
Categories |
 RSS Feed
RSS Feed
