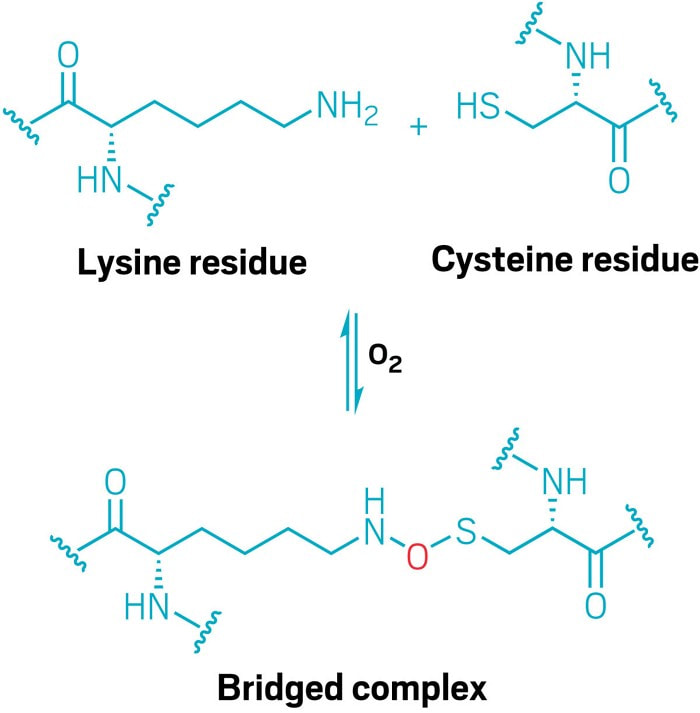
| The structure of proteins (and by extension their biological functions) is determined by post-translation modifications that give rise to a three dimensional structures from the peptide chain. One of the most common is the disulfide bond between two cysteine residues which is responsible for how curly human hair is and numerous other examples. Dr. Kai Tittmann at the Georg August University Göttingen recently discovered a new type of linkage between cysteine and lysine. While studying gonorrhoea bacteria they found that an enzyme lacked activity unless exposed to a reducing agent. Further investigation showed a previously undiscovered N-O-S bridge which produced a conformational change that rendered the enzyme inactive. The reducing agent breaks this bond and allows the enzyme to relax into the active state, therefore acting as a redox switch. A review of the Protein Data Bank shows this linkage in numerous organisms including humans and may represent a new target for drug discovery. |
|
0 Comments
|
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2021
Categories |
 RSS Feed
RSS Feed
